- Cite article
- Download PDF
- Share article
- 5 Downloads
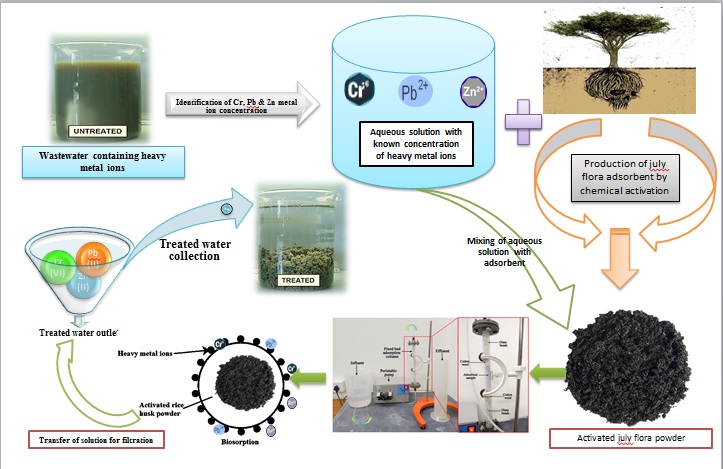
Adsorption of heavy metal ions (Cr, Pb & Zn) using prosopis julyflora roots has been investigated by batch adsorption and fixed bed column process. The various properties of adsorbent were analysed and the FT-IR spectra & SEM studies of prosopis julyflora powder, before and after adsorption of metal ions also examined. From the batch adsorption study, maximum amount of metal ion adsorption was found to be 87.12% for Cr (VI), 92.28% for Pb (II) and 95.62% for Zn (II) metal ions. The Freundlich isotherm model fitted well than the Langmuir adsorption isotherm with high regression values. From the column study, optimum bed height of 5 cm, flow rate of 5 mL/min and metal ion concentration of 100 mg/L was obtained by breakthrough analysis. The fixed bed column study followed Thomas & Yoon-Nelson model plots with good correlations and maximum desorption rate was achieved by adding 0.3M of concentrated H2SO4.