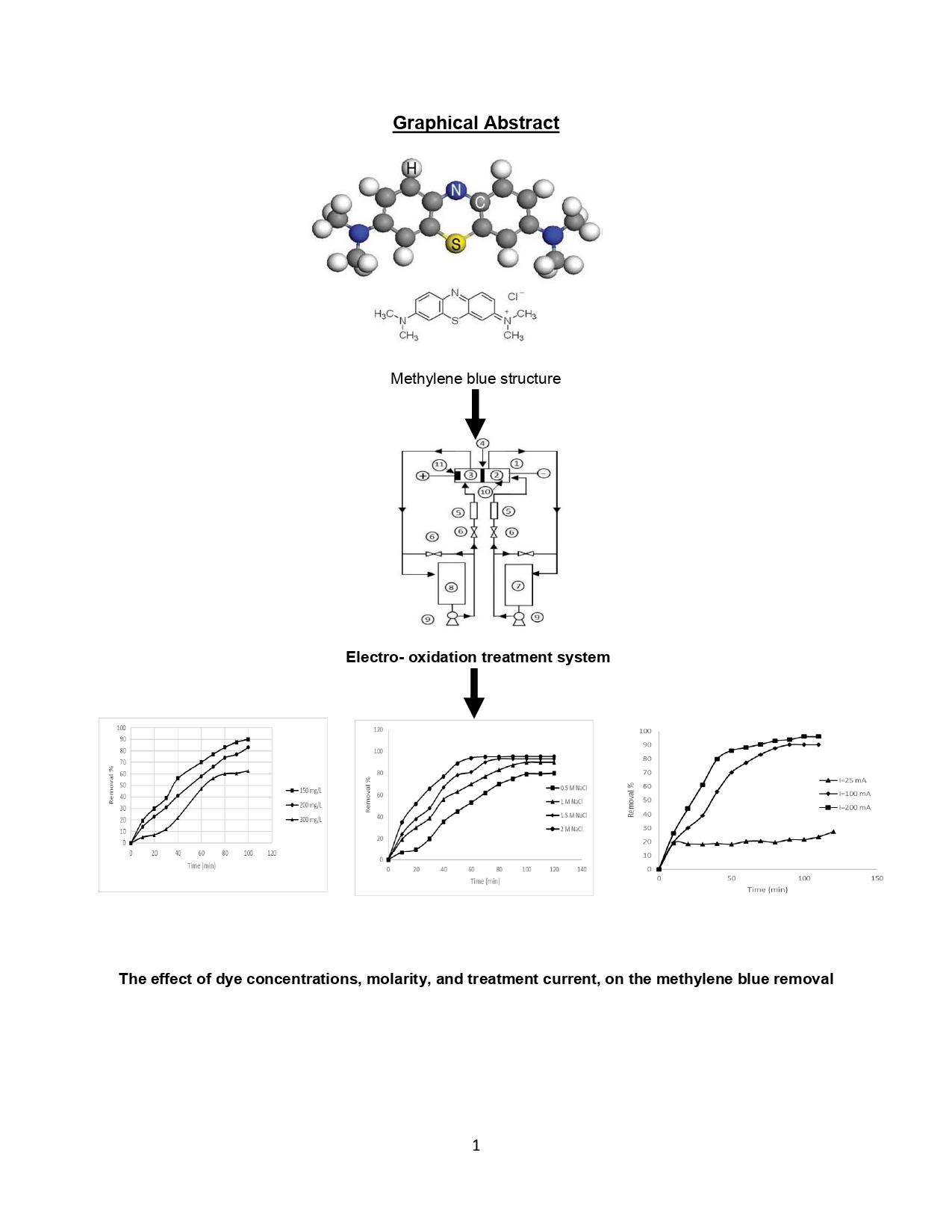
The objective of this study was to evaluate the efficiency of the electro-oxidation technique for the degradation of methylene blue from simulated wastewater. A flow-through electrochemical batch-circulation reactor was utilized to remove methylene blue from wastewater using a concentric tubes anode constructed of aluminum. The parameters studied in this work were dye concentration, electrolyte molarity, applied current, pH, and flow rate. The response indicators were the dye removal percentage and chemical oxygen demand (COD). The results demonstrated that the removal efficiency decreases as the dye concentration increases and also decreases with increasing pH. In contrast, the removal efficiency increases with increasing electrolyte molarity, flow rate, and applied current. The results clearly indicate that the applied process is effective for dye removal. The reactor operated in batch-circulation mode proved to be highly effective in removing the dye within a shorter time and with lower energy consumption.