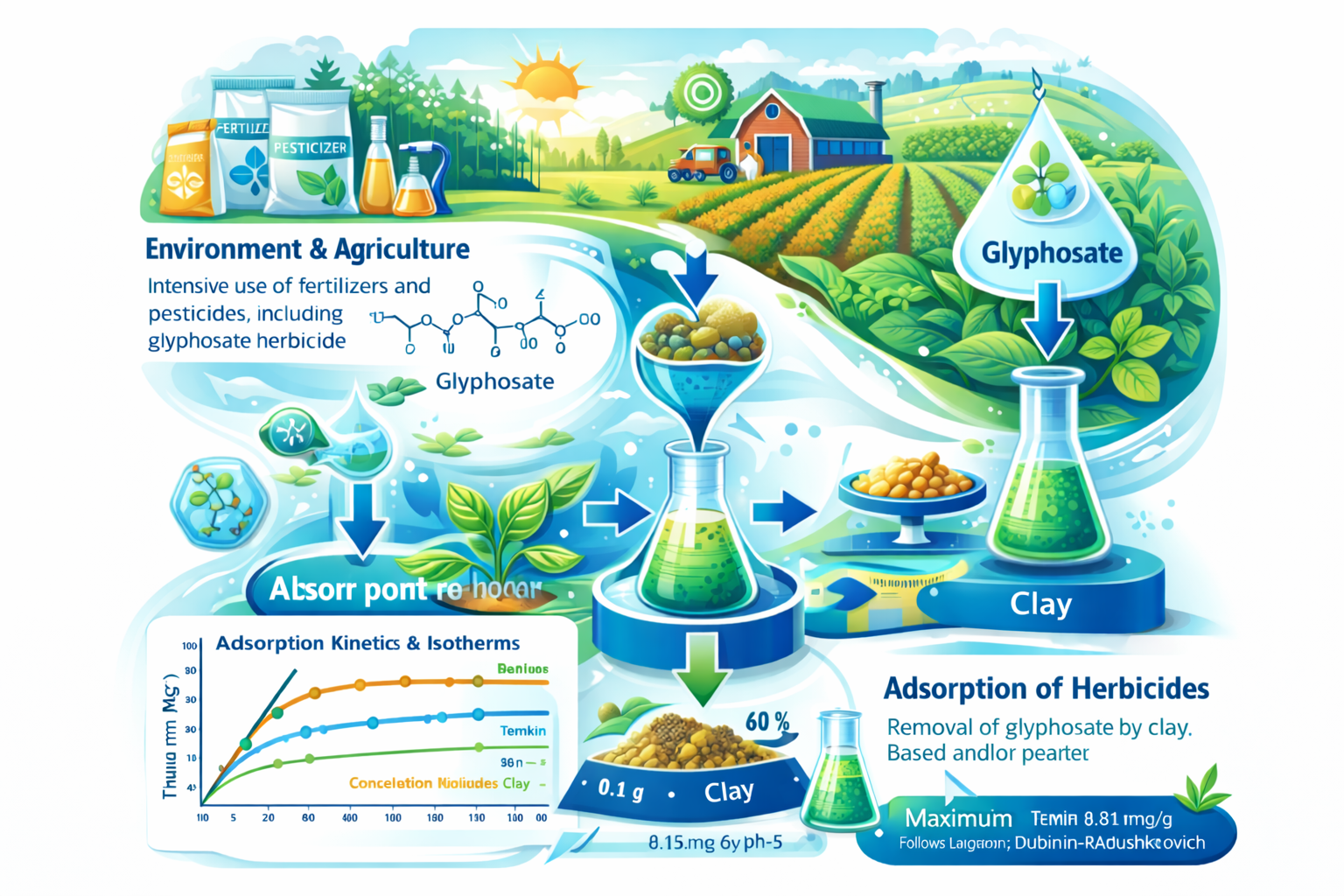
The natural environment is defined as the entirety of the Earth’s natural components and has been increasingly impacted by human activities in recent decades. Agricultural intensification, particularly in Algeria, has led to a substantial increase in the use of fertilizers and phytosanitary products to improve crop yield and quality. Among these products, glyphosate is the most widely used herbicide for weed control due to its effectiveness against annual, biennial, and perennial plants. This study investigates the removal of glyphosate from contaminated water using an abundant natural material, sodium-modified clay. Physicochemical characterizations were carried out to identify the surface functional groups and charge properties of the adsorbent. Adsorption experiments demonstrated that a clay mass of 0.1 g removed approximately 60% of glyphosate within 20 min at pH 5. Kinetic analysis showed that the adsorption process follows a pseudo-second-order model. Adsorption isotherm studies were conducted to elucidate the adsorption mechanism. The experimental data fitted well with the Langmuir, Temkin, and Dubinin–Radushkevich models, indicating monolayer adsorption with a maximum adsorption capacity of 18.18 mg g⁻¹. These results highlight the potential of sodium clay as an efficient, low-cost adsorbent for glyphosate removal from contaminated water.