- Cite article
- Download PDF
- Share article
- 18 Downloads
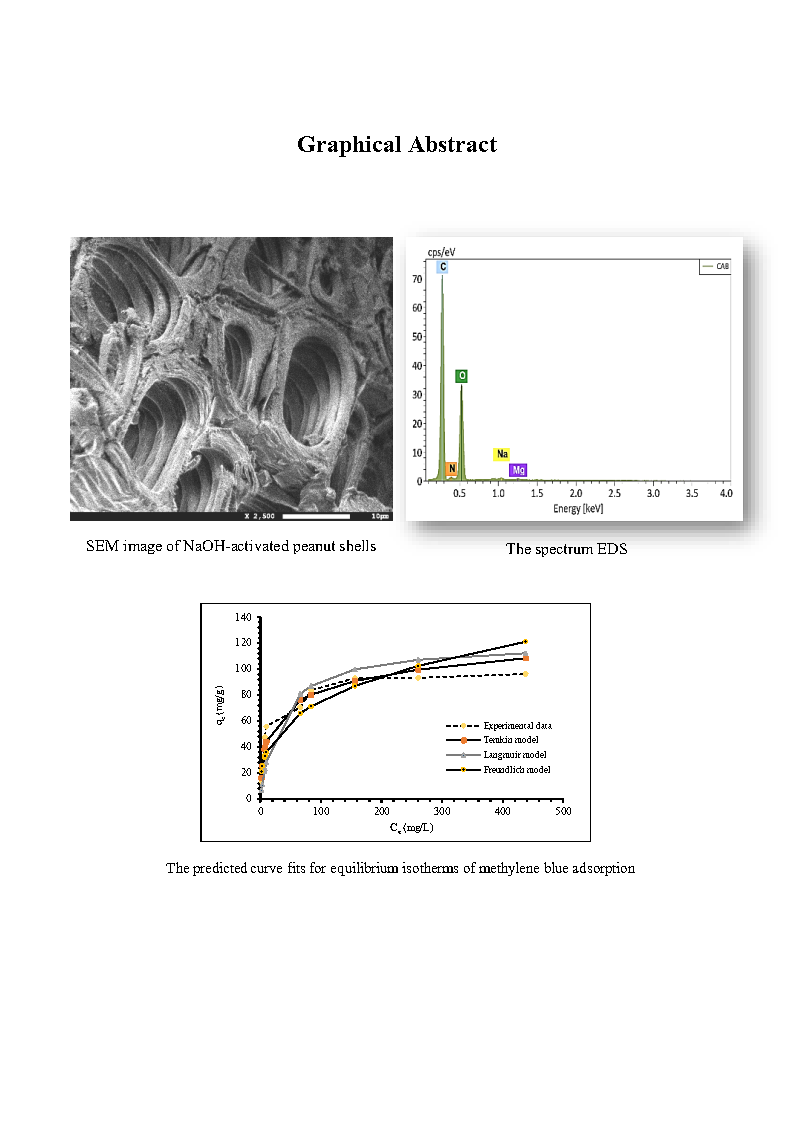
The main purpose of this study is to evaluate the possibility of using a lignocellulosic residue, peanut shells after alkaline activation, as a low-cost adsorbent for the removal of methylene blue dye from aqueous solutions. Adsorption experiments were conducted by varying parameters such as the initial pH aqueous solution, contact time, temperature, and initial concentration of adsorbate. Results show that pH has no apparent effect on methylene blue adsorption; more than 95% of methylene blue was removed. The adsorption capacity of methylene blue was slightly improved when the particle size of the adsorbent and the temperature decreased. A temperature uptake from 298 K to 328 K induced a decrease in the percentage of methylene blue removal from 91.36% to 84.31%. The kinetic data obtained at different concentrations were analyzed using pseudo-first-order and pseudo-second-order kinetic models. Also, the modeling of isotherm was considered by applying Langmuir, Freundlich, and Temkin models.
Kinetic and equilibrium data were best represented by pseudo-second-order and Langmuir models respectively according to determination coefficient R2 and error function values. The ultimate adsorption capacity reached approximately 100 mg/g. Thermodynamics findings revealed that the adsorption proposed is exothermic and spontaneous process. These results show that peanut shells can be a potential adsorbent for organic dyes removal from aqueous solutions.