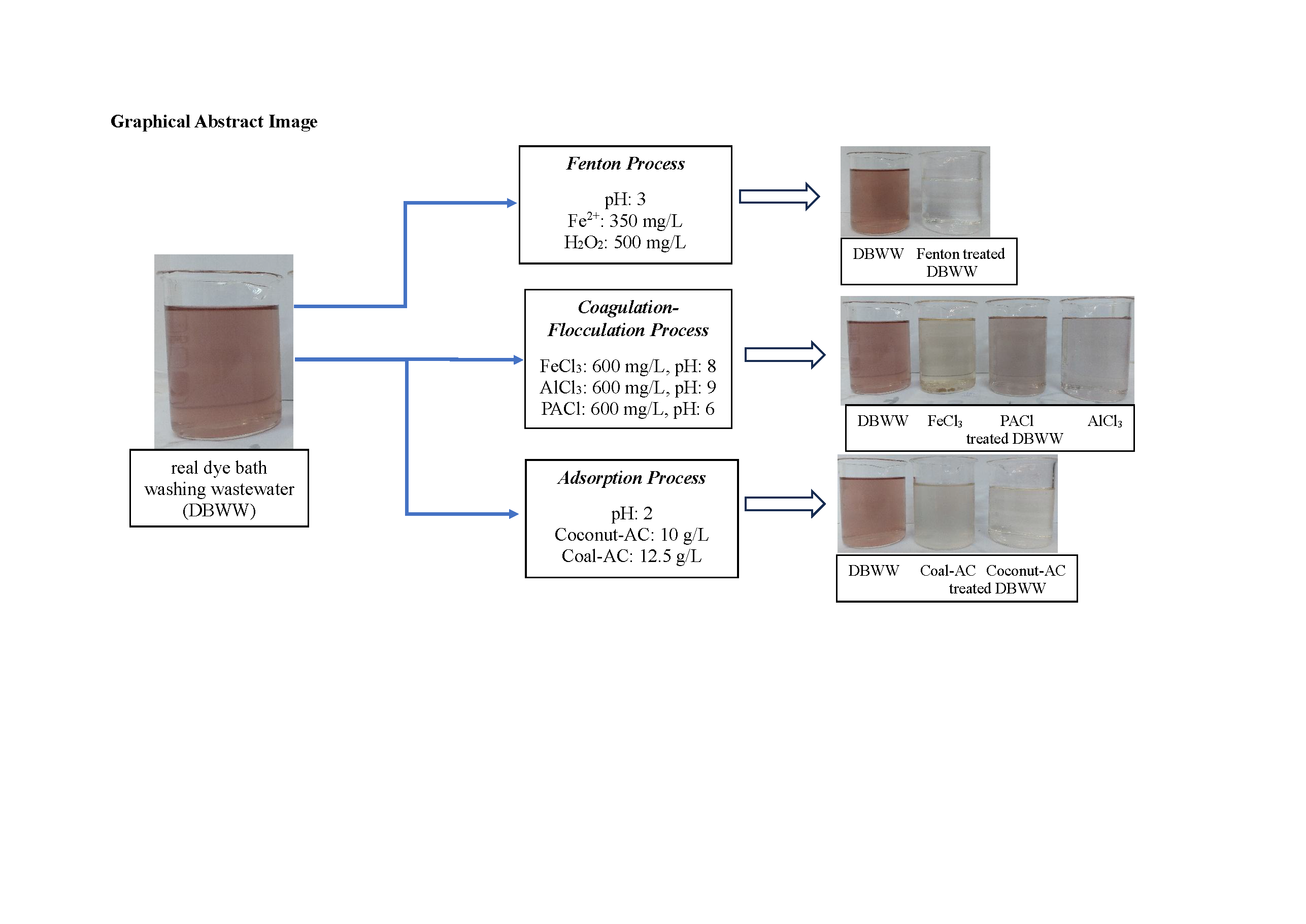
In this study, the real dye bath washing wastewater (DBWW) treatment using coagulation-flocculation, Fenton, and adsorption processes was investigated. The suitable pH and coagulant dose were researched using AlCl₃, FeCl₃, and PACl for the coagulation-flocculation process.The Fenton process was used to estimate optimum pH, oxidation time, and Fe2+ and H2O2. The optimum pH and adsorbent dose for activated carbon derived from coal and coconuts throughout the adsorption process were researched. COD removal efficiency followed the order PACl > AlCl3 > FeCl3, while color removal efficiency followed the order AlCl3 > PACl > FeCl3 in the coagulation-flocculation process. COD and color removals of 60.9% and 79.2% were reached at pH 6 and 600 mg/L PACl dose, respectively. 64.0% COD and 98.8% color removal were found at pH 3, 350 and 500 mg/L Fe2+ and H2O2 for Fenton process. Higher removal efficiencies were achieved using activated carbon obtained from coconut compared to activated carbon obtained from coal in the adsorption process. 69.7% COD and 76.3% color removal were obtained at pH 2, 10 g/L activated carbon dose and 1 h adsorption time. As a result, even though high COD removal was established in dye bath washing wastewater treatment employing three treatment methods, Fenton process produced high color removal