- Cite article
- Download PDF
- Share article
- 21 Downloads
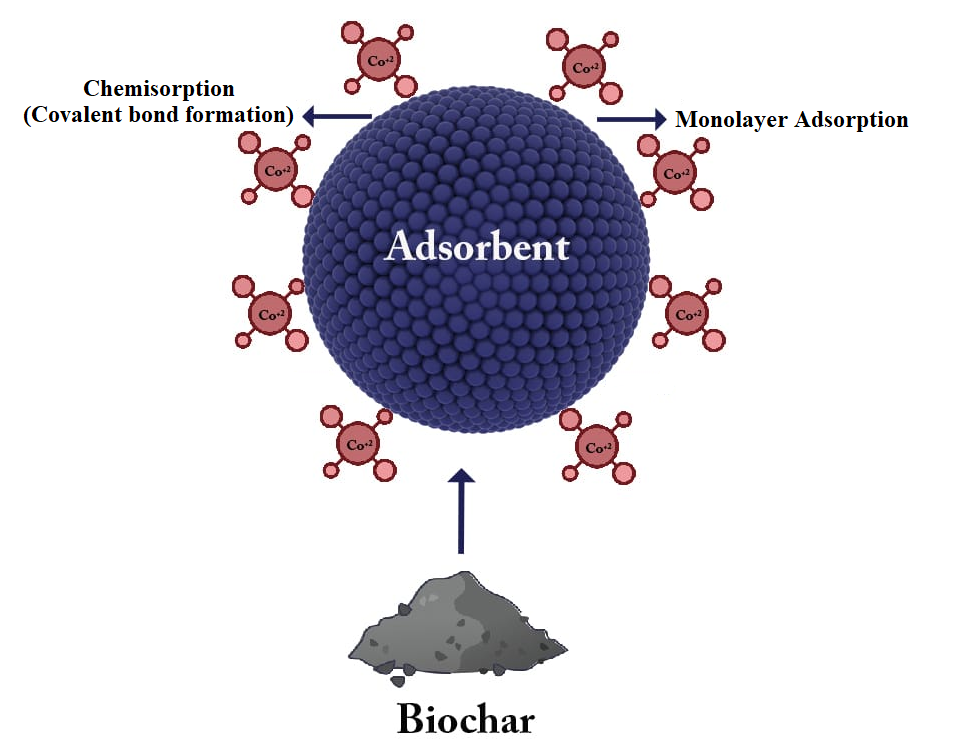
With an estimated volume of 162 thousand metric tonnes in 2020, India is the world's largest producer of tamarind. The country is home to a number of tamarind species, and production is spread out across the country. Farmers and agro-industries continue to encounter numerous challenges in the processing and disposal of waste created during tamarind harvesting and processing. However, because this agricultural waste is disposed of fast, it may pollute the ecosystem. As a result, this study focused on agricultural waste reuse for wastewater treatment, with tamarind seeds serving as the principal source for low-cost adsorbent (biochar) manufacture to remove harmful Co (II) ions rendered as an aqueous solution. Biochar is a form of charcoal produced by the pyrolysis of organic materials such as wood, agricultural waste, and animal manure. The surface characteristics of the synthesized biochar from tamarind seeds were investigated using FTIR spectroscopy, XRD, SEM, and EDAX. Tamarind seeds-based biochar (TSB) achieves maximal Co (II) ion adsorption at 9 pH, 50 °C, 90 min contact period, and 0.14 g/L adsorbent dosage. The Langmuir isotherm and Pseudo-second-order kinetics are the best-fitting isotherms and models for Co(II) ion adsorption using TSB. The maximal monolayer adsorption capacity (qm) of tamarind seeds-based biochar was determined by an adsorption isotherm analysis of 343.5 mg/g for Co (II) ions. Thermodynamic parameters successfully indicate that the current Co (II) ion adsorption utilizing TSB is possible, spontaneous, and endothermic. As a result, the adsorption investigation demonstrates that generated TSB has improved surface characteristics and capacity to remove Co (II) ions from wastewater effectively.