- Cite article
- Download PDF
- Share article
- 62 Downloads
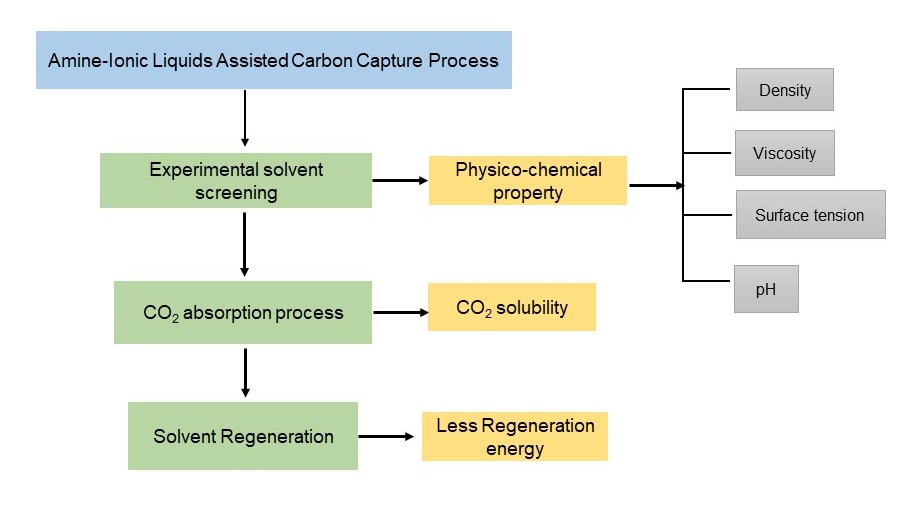
In recent years, there has been an increase in the earth's temperature due to the emission of CO2 which causes global warming. With a 1.9°C temperature increase predicted by the Intergovernmental Panel on Climate Change, reducing of CO2 is essential for all living species. Therefore, finding novel CO2-capturing strategies is the only way to eliminate this life threat. From post-combustion coal-fired power plants, CO2 separation is employed by a solvent-based chemical absorption method using Monoethanolamine (MEA) as standard solvent, which is the most practically proven and reliable process. The utilization of other solvents as absorbents in the CO2 absorption process such as amine solutions, ionic liquids (ILs), and amine-ILs solvent blend mixtures were reviewed. This review provides an overview of the CO2 capture process, emphasizing the importance of developing efficient and sustainable methods to mitigate greenhouse gas emissions. It also highlights the potential of using amine-ionic liquids mixtures as promising candidates for CO2 capture due to their unique properties for effective removal of CO2 by increasing CO2 absorption capacity as well as minimizing energy consumption during solvent regeneration.