- Cite article
- Download PDF
- Share article
- 47 Downloads
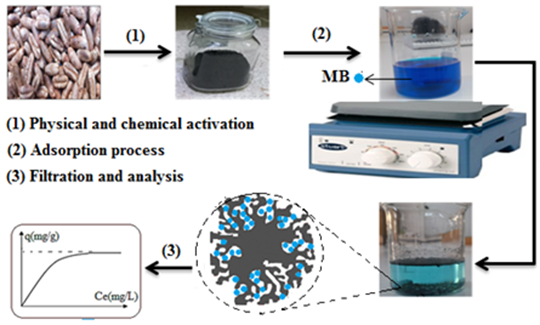
In this work, untreated activated carbon (UDP-AC) and activated carbon chemically treated with H₂SO₄ (TDP-AC) were successfully synthesized from date pits, a solid waste material. Characterization was performed on both UDP-AC and TDP-AC, and both samples were used to adsorb methylene blue (MB) dye from an aqueous solution. BET and BJH analyses were used to determine the surface area and pore size distribution. FTIR analysis confirmed the involvement of functional groups on the surfaces in the MB adsorption process. Scanning electron microscope (SEM-EDX) examination revealed that the adsorbents’ surfaces are heterogeneous and irregularly rough, with varying porosity, making them suitable for contaminant removal. The effects of several operating parameters, including adsorbent dose (0.05–0.5 g in 100 mL), initial pH (2–10), contact time (0–180 min), initial MB concentration (50–500 mg L⁻¹), and temperature (298–328 K), on MB removal were studied using batch mode operation. Adsorption kinetic data were best described by the pseudo-second-order model (R² = 0.99). The adsorption isotherm was well fitted by the Langmuir model, with maximum monolayer adsorption capacities of 270.27 mg g⁻¹ and 327.86 mg g⁻¹ at 25°C for UDP-AC and TDP-AC, respectively. Thermodynamic investigations indicated that the adsorption process is physical in nature, increases system randomness, and is feasible and spontaneous. These results provide evidence that activated carbon derived from date pits can serve as an inexpensive, eco-friendly, and efficient adsorbent for the removal of MB dye from wastewater.